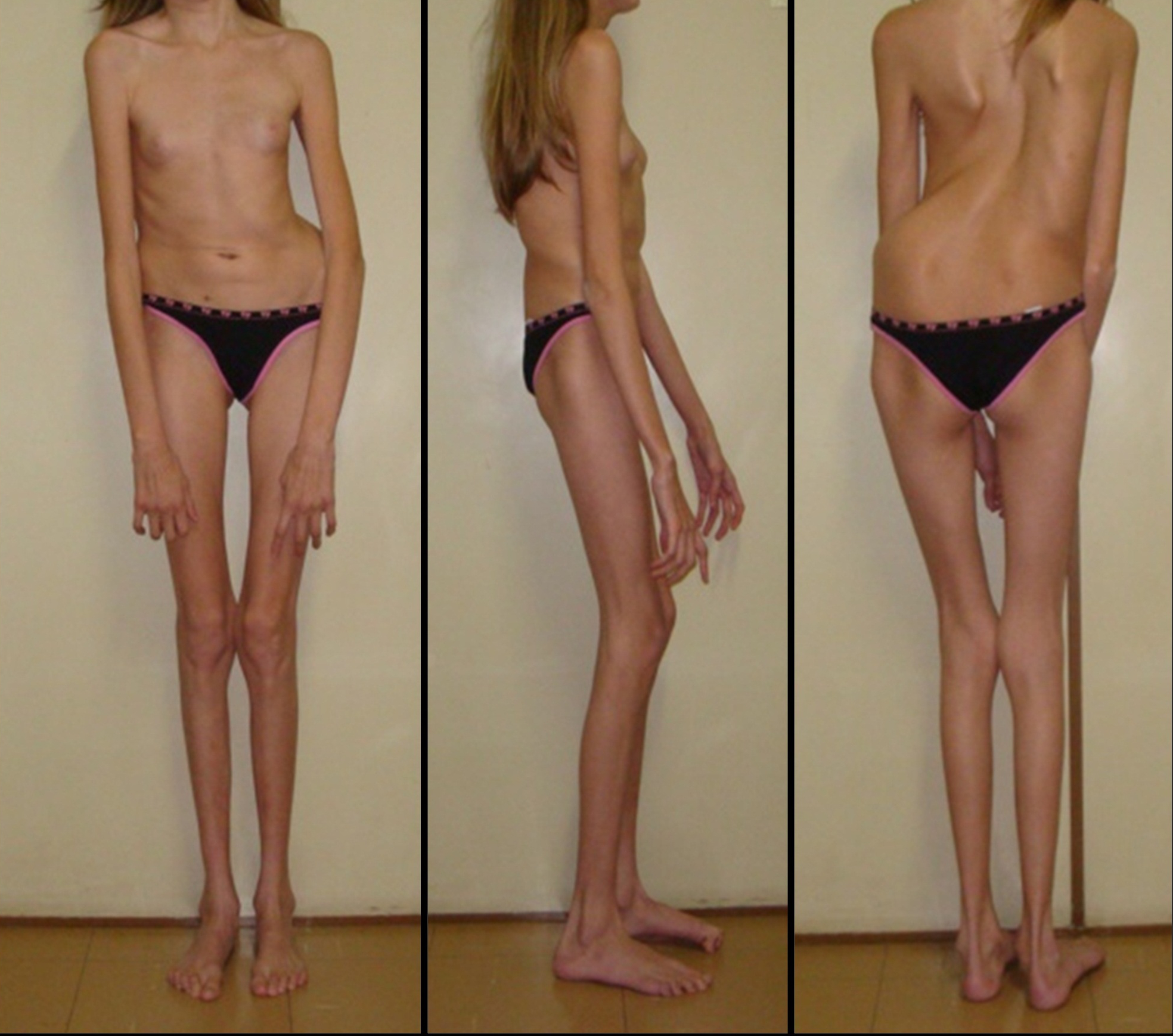
The proband (Figure ) is the first child of healthy parents. She was born at term, with a length of 50 cm and a weight of 3240 g. She walked at 18 months, and spoke at four years of age. She was however able to attend a regular school, but showed hyperactivity and difficulty to focus. She had a seizure at the age of 3 years, and a cardiac examination evidenced mitral insufficiency with a dystrophic valve. An evolutive scoliosis was noted at the age of 6 years, with progression.
At age 11, hypothyroidism was detected. She was referred at 13 years of age for a suspicion of MFS, showing: positive thumb and wrist sign, scoliosis, joint hyperlaxity, high-arched palate with dental crowding, dysmorphism and aortic root dilatation with dystrophic mitral valve. Ophthalmological examination revealed myopia and astigmatism but not ectopia lentis. A neuropsychological assessment showed global intellectual impairment (IQ 50) according to the Brazilian values of the Wechsler Intelligence Scale for Children-III (WISC-III), with major deficits in attention and executive skills. Thus, she met the Ghent criteria for Marfan syndrome, but also presented craniostenosis, hypothyroidism and intellectual deficiency. Conventional chromosome analysis was performed on phytohemagglutinin-stimulated lymphocytes from peripheral blood cultures, using GTG banding according to standard protocols.
Cell images were captured using the Ikaros Digital Imaging System (Metasystem, Altlussheim, Germany). G-banded analysis with a resolution of 550 bands per haploid karyotype revealed a female karyotype with a de novo balanced translocation involving chromosomes 6, 12 and 15, with breakpoints apparently at 6q22, 12q24 and 15q21 (Figure ). Figure 2 Cytogenetic and molecular data from the patient studied.
A) GTG-banded chromosomes showing the translocation involving chromosomes 6, 12 and 15. B) Array result for chromosome 15 showing the 1.9 Mb deletion (red) at 15q21.1 including the FBN1 gene (arrow). C) FISH with WCP probes of chromosomes 6, 12 and 15 in different color combinations showing a complex chromosomal rearrangement. D) FISH with probes RP11-631P6 (6q13) in red and RP11-46N22 (6q14.3) in green, showing signals next to each other on the normal chromosome 6 and separate signals on the der (6) chromosome.
E) FISH with probes RPRP11-627A9 (15q23) in green and RP11-793M16 (12q24.13) in red, showing signals of both on the derivative chromosome 12. F) Ideogram of the derivative chromosomes involved in the patient's complex chromosome rearrangement, showing the probes used to define the breakpoints and the 15q21.1 band deletion (arrow). Molecular Findings Genomic DNA was isolated from peripheral blood using a Gentra Puregene kit (Qiagen Sciences, Inc., Germantown, MD). Array study was performed with Affymetrix Cytogenetics Array 2.7 (Affymetrix Inc., Santa Clara, CA, USA) according to the manufacturer's instructions, using the Affymetrix Chromosome Analysis Suite software. Copy number state indicated an apparently contiguous interstitial deletion (1.9 Mb) on chromosome 15q21.1 in the region 45,466,733-47,335,104 bp (NCBI36/hg18) (Figure ).
Parental array analysis showed normal results. Molecular Cytogenetic Findings In order to better characterize the complex chromosome rearrangement, FISH was performed with region-specific BAC probes for chromosomes 6q, 12q and 15q. Clones were selected from the BACPAC Resource Center at the Children's Hospital Oakland Research Institute (Oakland, CA, USA) and prepared according to Guilherme et al.
FISH was performed with whole chromosome painting (WCP) probes for chromosomes 6, 12 and 15 (Cytocell, Cambrigde, UK). Individual and combined WCP probes were used in order to clarify the complex chromosomal rearrangement. Cell images were captured using the Isis Digital Imaging System (Metasystem, Altlussheim, Germany). FISH analysis revealed a quite complex rearrangement with eight breakpoints, as follows: 46, XX, t(6;12;15)(6pter→6q14::15q15.1→15q21.1::15q21.1→15q22.3::6q14→6q21::12q24.1→12qter;12pter→12q24.1::6q21→6q22.2::15q22.3→15qter;15pter→15q15.1::6q22.2→6qter)dn.arr 15q21.1(45,466,733-47,335,104)×1 (Figure and ). We report here on a girl with clinical features of the MFS spectrum and a 15q21.1 deletion including the entire FBN1 gene. This is the seventh study in the literature in which the deletion of FBN1 is confirmed by molecular techniques. Thus, to this date 18 patients with deletions including the whole FBN1 gene were reported, five of them belonging to the same family.
Interestingly, only 12 of these patients 2,7,11, present case present the typical MFS phenotype according to the Ghent criteria. Clinical variability in patients with different point mutations has been described, with patients with a nonsense mutation presenting milder phenotypes than patients with a missense mutation. Cases with a deletion can be compared to patients with a nonsense mutation, since the truncated mRNA in the latter cases is believed to be reduced, due to the nonsense-mediated decay (NMD) mechanism that prevents the expression of the truncated mRNA ,.
Thus, in cases of deletion and nonsense mutation, the MFS phenotype can result from FBN1 gene haploinsufficiency and, especially in cases of missense mutations, from a dominant negative effect. Hutchinson et al suggested that the clinical variability in MFS could be also due to variable FBN1 expression of the normal allele. The size of the deletions reported varies from small (less than 300 kb in 5 members of the same family) , including only the FBN1 gene, to large (up to 17.7 Mb). As found in our patient, when the deletion involves other genes besides FBN1, other unusual features can be found, such as those described here (craniostenosis, hypothyroidism and intellectual deficit). The patient described by Hiraki et al presented no sign of the MFS syndrome, probably due to her young age and severe clinical phenotype.
Our patient showed most of the skeletal features of MFS and an aortic root dilatation, but no ectopia lentis. Concerning the ocular system, she presented myopia and astigmatism.
Note: None of the art is made by me! Unnamed c 2d game for mac free. Sea2D (Sprite Engine Apple 2D) A 2D Game Sprite Engine written in C++ for macOS Written using at least Xcode 7.3 or greater on macOS 10.11. Was part of grad school individual research project.
Her deletion comprises 19 genes and predicted genes including FBN1, besides SEMA6D and COPS2 that may have contributed to the intellectual deficit and hypothyroidism, respectively. Of the 18 patients with a complete deletion of the FBN1 gene described so far, only seven were karyotyped: two presented normal karyotypes , , three had visible 15q deletions – , and one had a de novo translocation between the long arms of chromosomes 12 and 15 and a 4.9 Mb interstitial deletion at the translocation breakpoint of the long arm of chromosome 15 between the bands q21.1 and q21.2. Hilhorst-Hofstee et al performed karyotype analysis only in two patients, as part of the mental retardation screening. So, our patient is the first MFS case described presenting a complex chromosome rearrangement among chromosomes 6, 12 and 15. In the literature, up to 30-50% of the patients with a chromosomal rearrangement, both complex and reciprocal translocations, show an imbalance on the chromosomal or molecular level as an explanation for their phenotype.
The greater the number of breakpoints involved in a CCR, the greater the likelihood of genomic imbalances or position effect. Disruption of a gene could unmask a recessive mutation on the homologue allele, suggesting a greater chance for an abnormal phenotypic outcome. We emphasize the importance of using a combination of different molecular cytogenetic techniques in cases of chromosomal and/or genomic rearrangements involving the FBN1 gene, in order to better understand the extent of the molecular etiology of the Marfan syndrome and also to elucidate the genetic constitution of CCRs associated with diseases. Consent Written informed consent was obtained from the patient's parents for the publication of this case report and accompanying images. A copy of the consent form is available for review by the Editor-in-Chief of this journal. Authors’ original file for figure 2 Competing interests The authors declare that they have no competing interests. Authors' contributions MESC performed the molecular karyotyping and data analysis and wrote the manuscript; ABAP made the clinical evaluation of the patient; SST, TIM, ARND and RSG did the molecular analysis (FISH, WCP and SNP array); CBM performed a neuropsychological evaluation of the patient; LRJS made the cytogenetic analysis; and ABAP and MIM coordinated the study.
All the authors have read and approved the manuscript.
Text in this Example: Marfan Syndrome XY Father with Marfan syndrome XX Child with Normal mother YX Normal Child Marfan syndrome is a hereditary disorder passed from parent to child. Every person has two copies of every gene in the body (except some genes related to gender). One copy of each gene pair is inherited from each parent. It only takes one copy of the defective gene, inherited from one parent, for a person to have Marfan syndrome. If one parent of a couple has Marfan syndrome, each of their children has a 50 percent chance of inheriting the Marfan gene. X normal gene X defective gene Marfan syndrome is a disorder of connective tissue.
Connective tissue holds all the parts of your body together and helps control growth. Connective tissue gets some of its strength from a protein called fibrillin 1. Fibrillin 1 also plays an important role in controlling the growth and development of the body. In Marfan syndrome, the body produces fibrillin 1 that does not work properly. As a result, the connective tissue is not as strong as it should be, and the growth and development of the body are affected. About 3 out of 4 people with Marfan syndrome inherit the defective gene from a parent. In about 1 out of every 4 people with Marfan syndrome, the gene abnormality occurs due to a chance (spontaneous) mutation of the gene.
Source: National Heart Lung and Blood Institute, National Institutes of Health.
Copy of Ninguno - Created using PowToon - Free sign up at. Make your own animated videos and animated presentations for free. PowToon is a free tool that allows you to develop cool animated clips and animated presentations for your website, office meeting, sales pitch, nonprofit fundraiser, product launch, video resume, or anything else you could use an animated explainer video. PowToon's animation templates help you create animated presentations and animated explainer videos from scratch. Anyone can produce awesome animations quickly with PowToon, without the cost or hassle other professional animation services require.
I was diagnosed with in the summer of 2014 when I was only 13 years old. The effects of Marfan syndrome can be potentially life-threatening, so the diagnosis was devastating. There’s no way around that for me or for my family. However, I’ve come a long way since the initial shock and want to share my philosophies with others, whether their diagnosis is Marfan syndrome or something else. Marfan syndrome is not the end of the world! It is only the beginning of something new. When my cardiologist from Texas Children’s Hospital in Houston gave me the devastating news I could no longer play basketball, the game I loved and wanted to play in high school, college and maybe even in the NBA, I was crushed.
Jun 20, 2018 - You can also simply ping [yourenvironment].wpengine.com to find your. On a Mac, there is a very helpful tool for editing your hosts file called. Popular Alternatives to Winsock Packet Editor for Windows, Linux, Mac, Web, iPhone. Winsock Packet Editor (WPE) Pro is a packet sniffing/editing tool which is. Mar 12, 2014 - Networking, Routers, Switches, and Firewalls-WPE alternative for Mac OS X? Popular Alternatives to Packet Editor for Windows, Linux, Mac, Web, iPhone. Winsock Packet Editor (WPE) Pro is a packet sniffing/editing tool which is. Wpe alternative for mac.
However, I began looking for other sports I could play, like golf and tennis. I also chose to be the manager of my homeschool basketball team. As a result of having an open mind and trying something new, I now have a passion for sports that are safe for me to play the rest of my life (golf and tennis), and at the same time I can still be around basketball, the game I love.
Instead of playing, I am managing and learning from my coaches, as I now want to pursue a career in coaching. Don’t be alone on your journey with Marfan syndrome. At the time I was diagnosed, I did not know anyone with this disorder.
I felt scared and alone. As a college basketball fan, I knew, the former All-American basketball player from Baylor University and honorary NBA draft pick, had been diagnosed with Marfan syndrome two months prior to my diagnosis. So I decided to raise money for Isaiah’s new foundation to meet him and to help raise awareness for Marfan syndrome. That was the best decision I could have ever made. Isaiah and I text each other frequently.
He encourages me and I encourage him. He is like a big brother to me. I also attended the in Chicago this past summer. It was there that I made lifelong friends from Georgia, Pennsylvania, Ohio, California, Alaska and Montana. Closer to home, my doctor has introduced me to many teens my age in Houston who also have Marfan. Recently, six of us got together on a Friday night and hung out at Dave & Buster’s.
We are also going to an upcoming Houston Rockets game together and will participate in the Marfan Foundation’s first to raise money and awareness for Marfan syndrome. I also have a huge support system with my friends at church and my homeschool co-op. My best friend, Jonathan, has been to many doctor’s appointments with me and was at the hospital when I had eye surgery.
Marfan Syndrome Signs
I also have extended family from the staff at the Marfan Foundation. Don’t make Marfan syndrome or any setback your excuse. Make it your story.
When I was diagnosed with Marfan, I had a choice: I could either make my new life with Marfan syndrome my excuse or my story. So instead of being angry and turning inward with self-pity, I made the decision to encourage others with Marfan syndrome.
I am now focused on raising awareness of Marfan syndrome and money for the Marfan Foundation. I am the youth chair for the Houston Walk for Victory. My personal fundraising goal is $25,000 — and together, we hope to raise $100,000 in Houston. This wasn’t the path I chose, but it has certainly opened the door to my new purpose in life: to raise as much awareness and money as I possibly can and make Marfan syndrome a widely known disorder.
By doing so, many lives can be saved because people will know the signs. I also want to encourage and inspire anyone affected by Marfan syndrome or a related disorder or any setback in life. I believe life is too short not to live in victory.
Marfan Syndrome Chromosomal Mutation
Knowing this, I am going to make the very best of it. Owen (third from left) and friends. The Mighty is asking its readers the following: If you could go back to the day you (or a loved one) got a diagnosis, what would you tell yourself? If you’d like to participate, please send a blog post to Please include a photo for the piece, a photo of yourself and 1-2 sentence bio. Check out our page for more about our submission guidelines.
